
The highly attended New Drugs on the Horizon series at the AACR Annual Meeting 2026, organized by the AACR Chemistry in Cancer Research Working Group (CICR), featured first disclosures of the chemistry and biology of four novel anticancer molecules that are on the verge of clinical development.
The “New Drugs on the Horizon: Part 1” session, co-chaired by Lori S. Friedman, PhD, of ORIC Pharmaceuticals, and Lawrence G. Hamann, PhD, of Interdict Bio, highlighted new strategies to degrade target cancer proteins and selectively engage CD8+ T cells. Friedman was the 2024-2026 chair of the CICR Steering Committee.
“This is a flagship scientific series,” said Friedman, reflecting on the opportunity these sessions present to learn about new molecules and uncover breakthroughs in biology, understanding not only what the drug does, but what it reveals about the disease it targets. The hope, she concluded, is that many of these new molecules will turn into drugs that will ultimately benefit patients.
Related Coverage

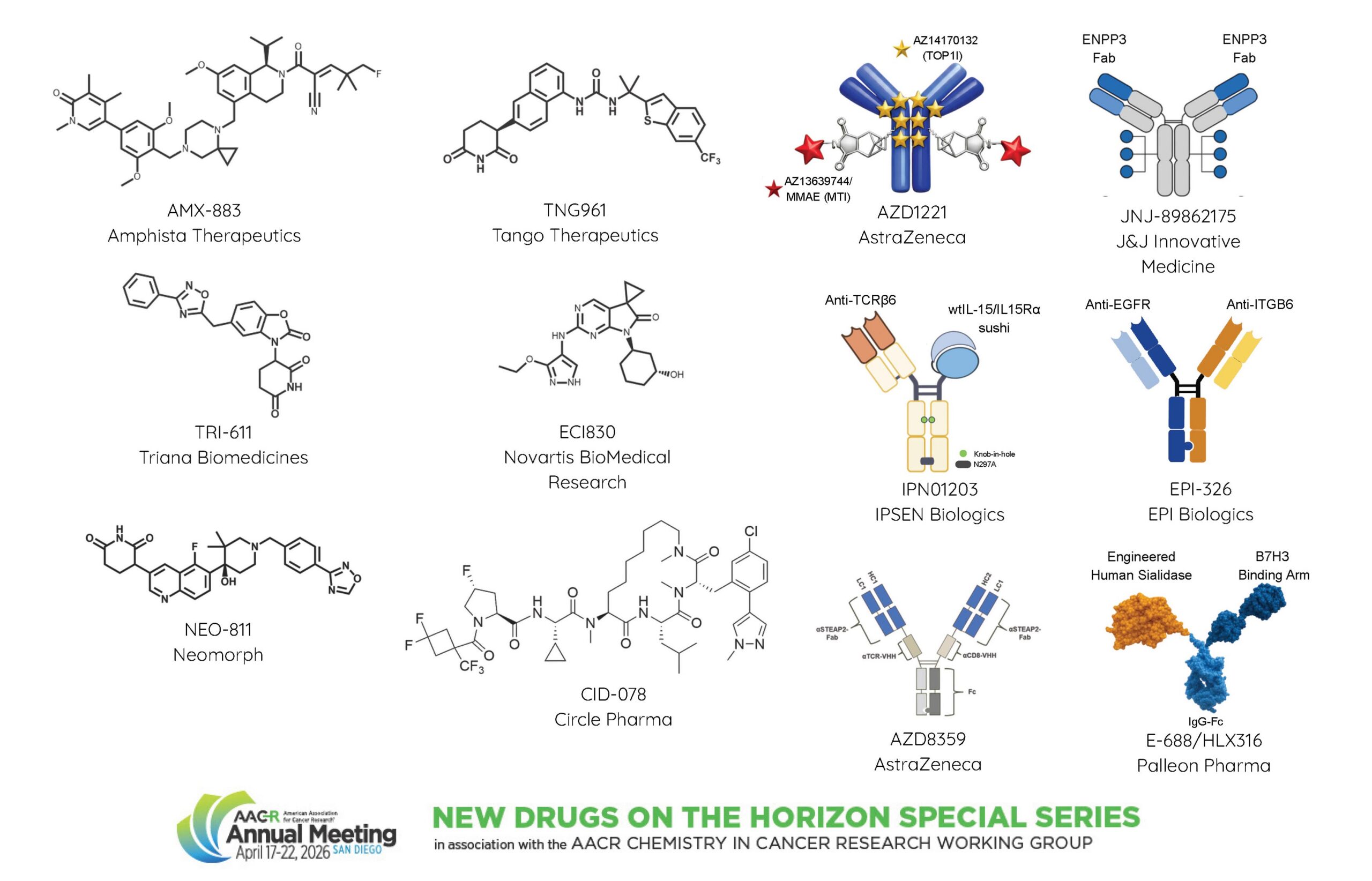
Philip Chamberlain, DPhil, of Neomorph, Inc., presented NEO-811, which is a potent and selective molecular glue degrader of ARNT (HIF-1β), a transcription factor involved in mediating cellular responses to low oxygen in the tumor microenvironment. NEO-811 is being developed for treatment of clear cell renal cell carcinoma (ccRCC), a disease characterized by a high prevalence of a mutation that causes loss of function of the VHL protein. This causes stabilization and constitutive expression of the HIF transcription factors, which in turn leads to transformation. ARNT is at the heart of the HIF signaling and was considered undruggable, Chamberlain noted, but is a great target for a molecular glue degrader.
He walked the audience through the screening and structure-based optimization steps that led to NEO-811. NEO-811 treatment caused potent and robust single-agent tumor regression in ccRCC xenografts and was effective in models that are resistant or refractory to HIF-2a inhibition. A phase I/II clinical trial is currently evaluating NEO-811 in patients with locally advanced or metastatic unresectable ccRCC.

Suzanne I. Sitnikova, PhD, of AstraZeneca, introduced AZD8359, a novel CD8-biased T-cell engager. Limitations of first-generation T-cell engagers in prostate cancer include the requirement of very specific tumor antigen targets and the occurrence of severe cytokine release syndrome events, which are mainly driven by CD4+ T cells, explained Sitnikova. Her team addressed these challenges by engineering AZD8359 with an anti-CD8 binding domain to preferentially activate CD8+ cells over CD4+ cells and by targeting the novel prostate cancer antigen STEAP2, which has limited expression in normal tissue and high expression across all stages of prostate cancer.
In vitro experiments confirmed that AZD8359 treatment mediated STEAP2-dependent, CD8-biased T-cell engagement and showed that AZD8359-treated CD8+ T cells have prolonged physical contact with tumor cells compared with CD4+ cells. In vivo treatment induced potent antitumor activity in mouse models. Sitnikova showed that AZD8359 caused reduced cytokine release than conventional T-cell engagers in vivo, translating to a wider preclinical therapeutic window. She added that promising results were also obtained through another CD8-biased T-cell engager design and that both candidates are being evaluated in clinical trials.
Hilary Elaine Nicholson, PhD, of Tango Therapeutics, highlighted TNG961, a first-in-class oral molecular glue degrader of HBS1L.

Nicholson explained that deletions in chromosome 9, which are frequent in many cancers, create a synthetic lethality opportunity through the loss of the focadhesin (FOCAD) protein that results in accumulation of stalled ribosomes. This renders FOCAD-deficient cells dependent on the HBS1L/PELO complex for ribosome recycling and survival.
By inducing selective degradation of HBS1L, TNG961 caused endoplasmic reticulum stress and activation of the unfolded protein response, resulting in loss of cell viability in FOCAD-negative cell lines and xenograft models from different tumor types. Notably, complete tumor regression was observed in models representing diseases with high unmet medical need, such as pancreatic and lung cancer. Furthermore, TNG961 showed a favorable safety profile in toxicology studies and presented opportunities for combination therapy with other inhibitors that selectively target MTAP deletion, which frequently co-occurs with FOCAD deletion. These data were concurrently published in Cancer Discovery, a journal of the American Association for Cancer Research. Having successfully completed preclinical characterization, TNG961 is ready for clinical development for the treatment of FOCAD-deleted cancers, Nicholson said.

Shyra J. Gardai, PhD, of EpiBiologics, described EPI-326, a tissue-selective EGFR-targeted bispecific antibody designed for the treatment of EGFR-driven cancers. The focus of most targeted protein degradation strategies is on intracellular targets, which leaves approximately 40% of the proteome unaddressed, said Gardai. The EpiTAC platform was developed for degradation of extracellular soluble and membrane proteins via bispecific antibodies that bind with one arm to pathogenic extracellular targets and with the second arm to degrader receptors.
Gardai highlighted how EGFR drives oncogenesis in multiple forms, through activating mutations, overexpression of the wild-type form, and by supporting HER2/HER3 signaling. “Currently, there are no therapeutics that can go after all these different forms of EGFR,” she said, while EPI-326 is able to target the protein agnostic to the form and could therefore have potential clinical application for different cancer types driven by different forms. Additionally, to improve safety and activity, the team selected ITGB6 as the degrader receptor because its expression is enriched in EGFR-responsive tumor types and limited within the healthy tissue. Upon binding, ITGB6 mediates internalization of EGFR, which is then degraded by the lysosome.
EPI-326 treatment caused tumor regressions in multiple EGFR-mutant non-small cell lung cancer (NSCLC) models, inducing a 90% complete response rate, and was also active in EGFR wild-type models. Notably, EPI-326 showed stronger antitumor activity than EGFR-targeted monoclonal antibodies in both EGFR-mutant and wild-type models. Combination treatment with either the EGFR tyrosine kinase inhibitor osimertinib (Tagrisso) or the KRAS inhibitor sotorasib (Lumakras) enhanced their antitumor activity in patient xenograft models of NSCLC and colorectal cancer, respectively. In toxicity studies, EPI-326 treatment was well tolerated with no EGFR-related toxicities. These favorable preclinical results allowed EPI-326 to enter clinical testing in a phase I trial in patients with EGFR-mutated tumors.
The recording of the full session is available on demand for registered Annual Meeting attendees through October 2026 via the virtual meeting platform.

More from the AACR Annual Meeting 2026 »
View a photo gallery of scenes from San Diego, join the conversation on social media using the hashtag #AACR26, and read more coverage in AACR Annual Meeting News and on Cancer Research Catalyst, the official blog of the AACR.





