
The New Drugs on the Horizon: Part 2 session spotlighted a human-sialidase-armed antitumor antibody, a novel CDK2 inhibitor, a molecular glue degrader, and a dual-payload antibody-drug conjugate (ADC).
Session Co-chair Danette L. Daniels, PhD, of Foghorn Therapeutics, said that the 12 presentations selected for the New Drugs on the Horizon series represent a mix of modalities, including biologics, small molecules, and macrocycles, and revealed a lot of breakthrough biology. Daniels is the 2026-2028 chair of the AACR Chemistry in Cancer Research Working Group (CICR), which sponsors the New Drugs on the Horizon series.
Related Coverage

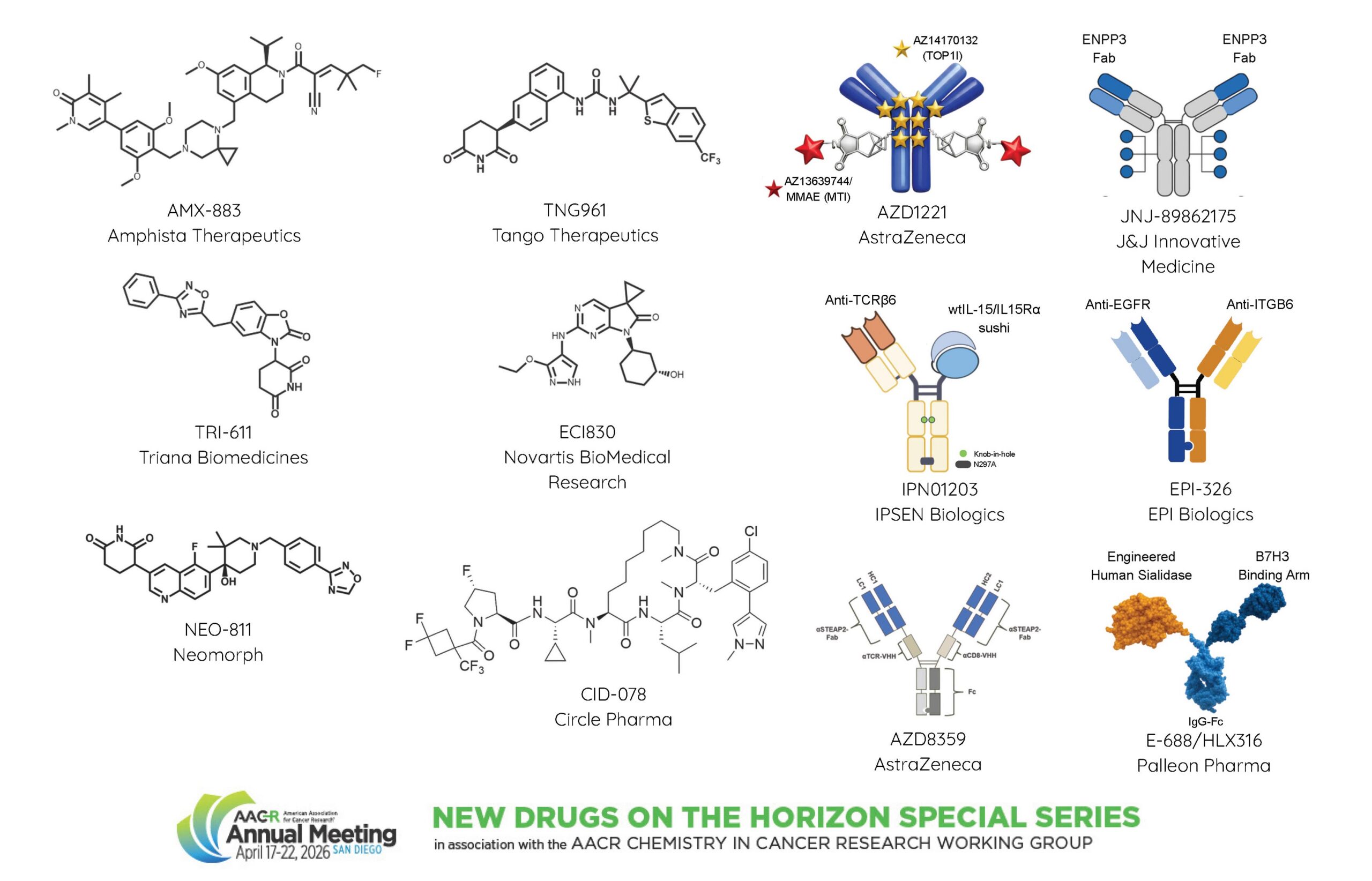
First presenter Jim Broderick, MD, of Palleon Pharmaceuticals, introduced HLX316/E-688, a first-in-class molecule that targets the immune checkpoint molecule B7-H3 and is armed with the human sialidase enzyme.
Hypersialylation promotes cancer development by suppressing innate and adaptive antitumor immunity. Initial clinical evaluation of a first-generation untargeted sialidase dimer allowed the researchers to establish proof of mechanism and identify key limitations that hindered efficacy. This experience led to the development of HLX316/E-688 as a tumor antigen-targeted sialidase that retained in vivo tumor desialylation activity for more than seven days and showed improved preclinical activity compared with the untargeted dimer.
Broderick pointed out that B7-H3 expression and hypersialylation are both present on a variety of tumor types, suggesting broad potential of HLX316/E-688 across multiple cancer patient populations. A phase I trial of HLX316/E-688 is initiating in platinum-resistant ovarian cancer, which shows high expression of B7-H3 and hypersialylation.

Zainab Jagani, PhD, of Novartis Institutes for BioMedical Research, Inc., discussed ECI830, a CDK2 inhibitor, for the treatment of certain HR-positive, HER2-negative breast cancer patients.
Targeting cell cycle through CDK4/6 inhibitors, in combination with endocrine therapy, represents a clinically proven strategy for HR-positive breast cancer, yet therapy resistance and progression remain a challenge, said Jagani. In particular, patients whose tumors express high levels of cyclin E have lower progression-free survival compared with patients with lower expression, and cyclin E levels increase after treatment. Jagani and team hypothesized that combined inhibition of CDK4 and CDK2, which works in complex with cyclin E, may yield more durable responses. They developed ECI830 as a potent and highly selective ATP-competitive CDK2 inhibitor that enhanced the effects of the CDK4/6 inhibitor ribociclib (Kisqali) in breast cancer cells and tumor models. Jagani added that ECI830 showed efficacy as a monotherapy in CCNE1-amplified ovarian and lung cancer models.
ECI830 is currently being evaluated in a phase I clinical trial as a single agent and as part of a combination in patients with advanced HR-positive, HER2-negative breast cancer and other advanced solid tumors.

Vito J. Palombella, PhD, of Triana Biomedicines, presented TRI-611, a selective molecular glue degrader of the ALK receptor tyrosine kinase that is expressed as a fusion protein in approximately 5% of cases of non-small cell lung cancer (NSCLC). Multiple tyrosine kinase inhibitors have been approved for the treatment of ALK-positive NSCLC, but chemotherapy remains the only option after progression, said Palombella. TRI-611 was designed to facilitate ALK ubiquitination and degradation through a structural motif called degron.
TRI-611 treatment induced dose-dependent ALK degradation and tumor shrinking in models of NSCLC and was active against mutated forms of the EML4-ALK fusion protein that are associated with resistance to tyrosine kinase inhibition. Furthermore, TRI-611 caused tumor regression in an ALK-positive patient-derived tumor model and in intracranial tumor xenografts, highlighting its potential for the treatment of central nervous system metastases. Palombella emphasized that, due to its high selectivity and preclinical safety, TRI-611 may allow combinations with other therapeutics.
TRI-611 is being studied in a phase I trial and has received Fast Track designation from the U.S. Food and Drug Administration.

Megan Rice, MS, of AstraZeneca, highlighted the discovery and development of a CD30 dual-payload ADC (dpADC). Brentuximab vedotin (Adcetris) was the first CD30-targeting ADC to be approved for the initial treatment of Hodgkin lymphoma and anaplastic large-cell lymphoma (ALCL). Unfortunately, many patients relapse after treatment. DpADCs enable simultaneous delivery of two different drugs into the same cancer cells to drive more robust tumor responses, overcome drug resistance, and address tumor heterogeneity, explained Rice. CD30 dpADC combines a microtubule inhibitor and a topoisomerase I inhibitor.
In vitro experiments showed that CD30 dpADC achieved specific binding to CD30-positive cells, was efficiently internalized, induced payload-driven DNA damage and cell death. Treatment with CD30 dpADC showed preclinical activity both in Hodgkin lymphoma and ALCL tumor models. Furthermore, because of reduced free-payload accumulation, CD30 dpADC treatment at clinically relevant doses demonstrated improved tolerability compared with brentuximab vedotin.
Planning of a phase I clinical trial of CD30 dpADC is underway.
The recording of the full session is available on demand for registered Annual Meeting attendees through October 2026 via the virtual meeting platform.

More from the AACR Annual Meeting 2026 »
View a photo gallery of scenes from San Diego, join the conversation on social media using the hashtag #AACR26, and read more coverage in AACR Annual Meeting News and on Cancer Research Catalyst, the official blog of the AACR.





